Carbohydrates and its Classification
Carbohydrates Definition
Carbohydrates word is made up of two words “carbo” meaning “carbon” and “hydrate” meaning “water or H2O”. In short, carbohydrates consist of carbon (C), hydrogen (H), and oxygen (O) elements in a 1:2:1 ratio with the general formula of Cx(H2O)y.
In the formula, x can be a whole number from three to many thousands, while Y can be the same whole number as X e.g., glucose molecule (C6H12O6) or it may be a different whole number as in sucrose (C12H22O11).
Carbohydrates can be polyhydroxy aldehydes or polyhydroxy ketone. It is called polyhydroxy due to the presence of many hydroxyl groups (-OH) in its structure. When carbohydrates contain an aldehydes group (HC=O) in its structure it is called aldo sugar and if the ketone group (R2C=O) is present then it is called keto sugar.
Properties of Carbohydrate
Carbohydrates are important biological molecules due to their important properties such as:
- They are abundant in all living organisms.
- Green plants are the main source of carbohydrates.
- Carbohydrates are present in vegetables, fruits, paper, grains, wood, sugar cane, cotton, and milk sugars.
- They are the key source of energy for the body.
- They play structural and functional roles in humans, animals, and plants.
- Carbohydrates are the main constituents of cell walls, thus providing support and protection to plant cells.
- Carbohydrates combine with other biological molecules to make a variety of useful compounds. For example, glycoproteins and glycolipids are formed when carbohydrates combine with proteins and lipids, respectively.
Video lesson on Carbohydrates Definition
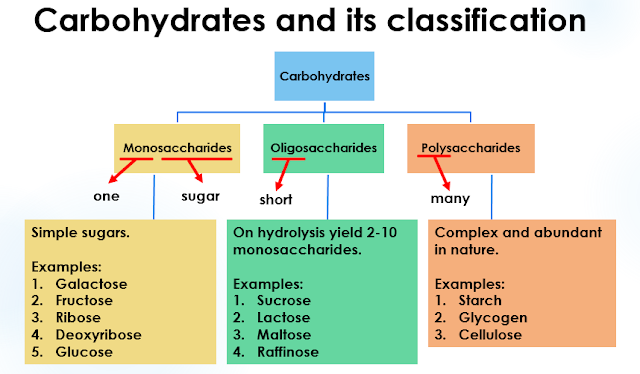
Classification of Carbohydrates
Carbohydrates are classified into three categories based on the number of sugars present in them. The three main classifications of carbohydrates are:
- Monosaccharides
- Oligosaccharides
- Polysaccharides
|
| Carbohydrates and its classification |
1. Monosaccharides
Monosaccharides word is made up of two words i.e., mono and saccharides, where “mono” means “single” and “saccharides” means “sugar”. So, monosaccharides are carbohydrates that consist of only one sugar molecule.
Monosaccharides are simple sugar, sweet in taste, and soluble in water. They cannot be broken down or hydrolyzed into simpler sugars. Their general formula is (CH2O)n, where n represents the number of carbon atoms ranging from 3 to 7. The monosaccharides with three, four, five, six, and seven carbon atoms are called trioses (3C), tetroses (4C), pentoses (5C), hexoses (6C), and heptoses (7C), respectively.
In monosaccharides, all carbon atoms have a hydroxyl (-OH) group, except one carbon. Moreover, they may be aldo-sugar or keto-sugar depending on the presence of aldehyde or ketone group in their structure.
In solution, monosaccharide having carbon atoms 4 or more seldom switches from open-chain structure to stable ring structure by making the bond between the carbonyl group and one hydroxyl group of the same molecule.
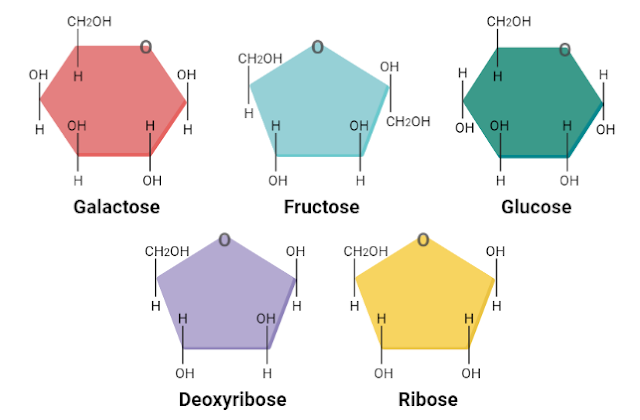
Examples of Monosaccharides
Some important examples of monosaccharides are:
- Galactose
- Fructose
- Ribose
- Deoxyribose
- Glucose
|
| Examples of monosaccharides |
1. Galactose
It is an aldohexose sugar and sometimes abbreviated as “Gal”. It is as sweet as glucose sugar with the same molecular formula of C6H12O6. It is present in honey, celery, cherries, corn, and beets. Galactose is the main component of glycolipids present in the brain and myelin sheath of neurons, thus it is also called “brain sugar”.
2. Fructose
Fructose is a ketohexose sugar. It is also known as fruit sugar because it is mainly present naturally in many fruits such as sugar beets, honey, fruits juices, and some vegetables. The molecular formula of fructose is C6H12O6.
3. Ribose
It is an important aldopentose sugar. It can be obtained from beef, cheese, mushrooms, milk, egg, and poultry. Its molecular formula is C5H10O5.
It performs many important functions in the cell. It is an important component of ribonucleic acid (RNA), where it alternates with the phosphate group to make a backbone of RNA polymer. Moreover, it is also present in adenosine triphosphate (ATP) that provides energy to the body. Ribose sugar forms a cyclic structure in a solution known as ribofuranose.
4. Deoxyribose
It is a pentose sugar. It is present specifically in DNA molecules, where it alternates with the phosphate group to form a DNA backbone. It is called “deoxy” because it is derived from ribose sugar by the loss of one oxygen molecule. This structural change allows it to help in the replication of DNA molecules in a double helix configuration. The molecular formula of deoxyribose is C5H10O4.
5. Glucose
The most important monosaccharide in a living cell is glucose. It is an aldohexose sugar and is present in potatoes, bread, and fruits such as grapes, figs, and dates. In plants, glucose is synthesized during the process of photosynthesis.
During photosynthesis, in the presence of sunlight, plants absorb water from the soil and carbon dioxide from the air to prepare glucose and oxygen as a by-product. About 10g of glucose stores 717.6Kcal of solar energy in the form of chemical energy. So, it is the major source of energy for animal and plant cells and serves as fuel for the body.
In blood, about 0.8% glucose is present and its amount is controlled by insulin hormone produced by the pancreas.
Glucose molecules combine with each other or with other monosaccharides to form many oligo and polysaccharides (starch, glycogen, cellulose).
The cyclic form of glucose is known as glucopyranose and its molecular formula is C6H12O6.
2. Oligosaccharides
In the word Oligosaccharides, “oligo” means “short” and “saccharides” = “sugar”. Oligosaccharides are carbohydrates that are made up of a small number of monosaccharides ranging from 2 to 10 sugars. They are less sweet, less soluble in water, and can be hydrolyzed or broken down into 2-10 simpler sugars.
Oligosaccharides are formed when two or more monosaccharides form a bond with each other. The bond between two monosaccharides is called a glycosidic bond in which the oxygen from a hydroxyl group becomes linked to the carbonyl carbon with the release of one water molecule. Disaccharides are formed when two sugars combine, while trisaccharides are formed when three sugars are combined by glycosidic bonding.
Examples of Oligosaccharides
The main examples of oligosaccharides are:
- Sucrose
- Lactose
- Maltose
- Raffinose
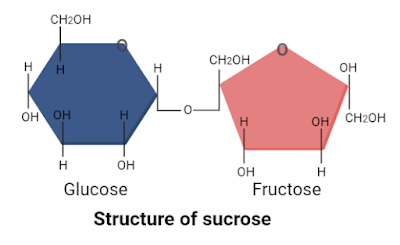
1. Sucrose
Sucrose, also known as “cane sugar” or “table sugar”, is a disaccharide. It is formed by the joining of one molecule of glucose and one molecule of fructose through glycosidic bonding. The molecular formula of sucrose is C12H22O11. Sucrose can naturally be obtained from sugar beets, sugarcane, dates, sugar maple sap, and honey.
|
| Structure of sucrose |
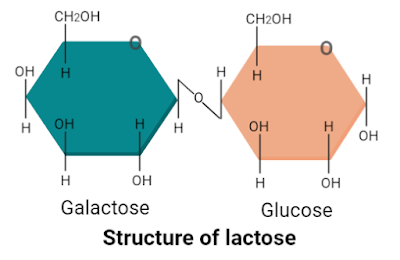
2. Lactose
Lactose is a disaccharide that is made up of one molecule of glucose and one molecule of galactose. The molecular formula of sucrose is C12H22O11. It is also known as “milk sugar” because it is naturally present in milk only. People who lack lactose digesting enzyme lactase suffer from lactose tolerance.
|
| Structure of lactose |
3. Maltose
Maltose is formed when two glucose molecules join with each other through α1-4 glycosidic linkage, thus it is also a disaccharide. Grains, cereals, sweet potatoes, and some fruits are rich sources of maltose. Its molecular formula is C12H22O11.
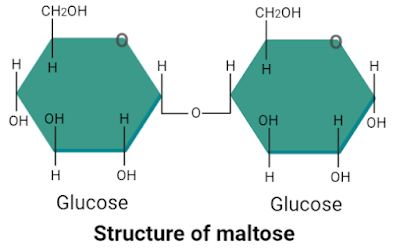
|
| Structure of maltose |
4. Raffinose
The trisaccharide raffinose is made up of galactose, glucose, and fructose. It can be broken down into sucrose and galactose by the α-galactosidase enzyme. Humans and other monogastric animals cannot digest this sugar due to the absence of α-galactosidase enzyme, so it passes through the stomach and small intestine as such. However, in large intestines, bacteria help in their digestion due to their ability to produce the α-galactosidase enzyme. Beans, brussels sprouts, cabbage, broccoli, asparagus, various vegetables, and whole grains are the main sources of this sugar.
3. Polysaccharides
Polysaccharides (Poly = Many and saccharides = sugar) are the most abundant and complex sugar in nature. They are made up of a large number of monosaccharides joined together either in a branched structure or unbranched structure. They are tasteless, insoluble in water, have high molecular weight, and can be obtained from animals and plants.
Examples of Polysaccharides
The three main examples of polysaccharides are:
- Starch
- Glycogen
- Cellulose
1. Starch
Starch is a polymer of α-glucose molecules. It can be found in tubers, potatoes, fruits, and seeds. In plants, it is used to store excessive glucose in the form of food and can be utilized when needed. Starch provides energy to the animals by yielding glucose molecules on hydrolysis. It can easily be broken down with the help of the amylase enzyme present in saliva.
Starch can be dissolved in warm water. An iodine test can be performed to identify starch. Starch gives blue color with iodine. The molecular weight of starch is variable and depends on the number of branches attached.
The arrangement of α-glucose molecules divides starch into two forms i.e., amylose and amylopectin.
In amylose, glucose molecules are arranged in an unbranched structure by forming α 1-4 linkage between C1 of one glucose molecule and C4 of another glucose molecule.
On the other hand, amylopectin a is a branched structure due to the formation of two types of linkages i.e, α 1-4 and α 1-6 linkages. The α 1-6 linkage is formed between C1 of one glucose molecule of unbranched chain and C6 of another glucose molecule of another chain, thus adding a branch to the unbranched structure.
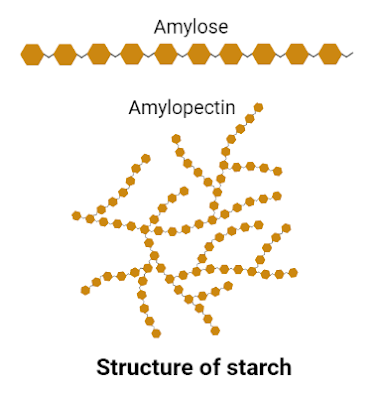
|
2. Glycogen
Glycogen is known as “animal starch” because it is found in the liver, muscles, and other cells of animals. It helps in the storage of carbohydrates in an animal's body. It is a polymer of α-glucose molecules. Just like starch, it also has α 1-4 and α 1-6 linkages among glucose molecules, thus forming a highly branched structure. The molecular weight of glycogen is 666.6 g/mol.
It is poorly soluble in water, gives a red color with iodine, and can easily be broken down into glucose molecules. The two important enzymes required for glycogen break down are glycogen phosphorylase and glycogen debranching enzyme.
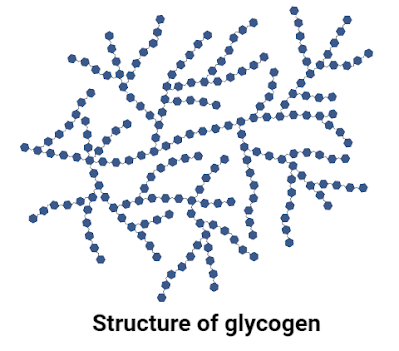
|
3. Cellulose
Cellulose is the most abundant polysaccharide in nature. It is the main constituent of the cell wall providing support and protection to plant cells. Cotton is the pure form of cellulose.
It is a polymer of β-glucose molecules arranged in an unbranched structure by β 1-4 glycosidic linkage. Cellulose is tasteless, odorless, cannot be dissolved in water, and gives no color with iodine. Its molecular weight is 162.1406 g/mol.
Cellulose cannot be digested in the digestive tract of humans due to a lack of cellulose digesting enzyme called cellulase. Herbivores, on the other hand, have a special consortium of bacteria that synthesize cellulase enzymes and help in the digestion of cellulose present in feed.

|
Video Lesson on Classification of Carbohydrates
Some Questions and Answers
1. Give three examples of monosaccharides.
A. Glucose, fructose, and galactose are three examples of monosaccharides.
2. State two forms of starch?
A. Amylose and amylopectin are two different forms of starch.
3. Is cellulose a branched structure?
A. No, cellulose is not a branched structure because it does not contain β 1-6 glycosidic linkage.
4. What are the 3 classifications of carbohydrates?
A. Carbohydrates are classified into monosaccharides, oligosaccharides, and polysaccharides.
5. What do starch, glycogen, and cellulose have in common in terms of their molecular makeup?
A. These three polysaccharides are made up of long chains of glucose molecules joined with each other through glycosidic linkage.
6. What is the similarity and difference between glycogen and starch?
A. They both have α-glucose as their subunit, are branched structure, and have α 1-4 and α 1-6 linkages among glucose molecules.
7. Why humans cannot digest cellulose?
A. Cellulose can be digested by cellulase enzyme which is absent in the digestive tract of humans.
8. Which test can be used to detect starch?A. Iodine test is used to detect starch. Iodine turns blue if starch is present in the sample.




0 Comments